The EDC-Free Europe coalition, of which HEAL is a member, has launched a new infographic illustrating the harmful impacts of exposure to known and suspected endocrine disruptors on our health and the environment.
Scientific evidence of the health-harming impacts of exposure to endocrine disrupting chemicals (EDCs) has been piling up for years. Regrettably, there are presently no harmonised criteria to identify such chemicals across sectors, uses and legislations in the European Union. HEAL’s new infographic illustrates how the upcoming revision of the EU Classification, Labelling and Packaging (CLP) Regulation can help solve this problematic loophole within European chemicals laws.
The Classification, Labelling and Packaging (CLP) Regulation sets out how the EU classifies and communicates about the hazardous properties of chemical substances and mixtures. Introduced in 2008, CLP has created a single harmonised process for the identification and labelling of the harmful properties of substances, which applies across all sectors. It allows all users of chemicals – from companies across supply chains to workers and consumers – to find out about their potential hazardous properties.
However, in its current form CLP has several important limitations, starting with the fact that it does not cover all the hazards that are relevant to our health and the environment, such as endocrine disruption.
The 2020 Chemicals Strategy for Sustainability committed to fixing these flaws by revising the CLP legislation and introducing new hazard classes. This is particularly important when it comes to the identification of endocrine disruptors, which needs to be fastened and improved.
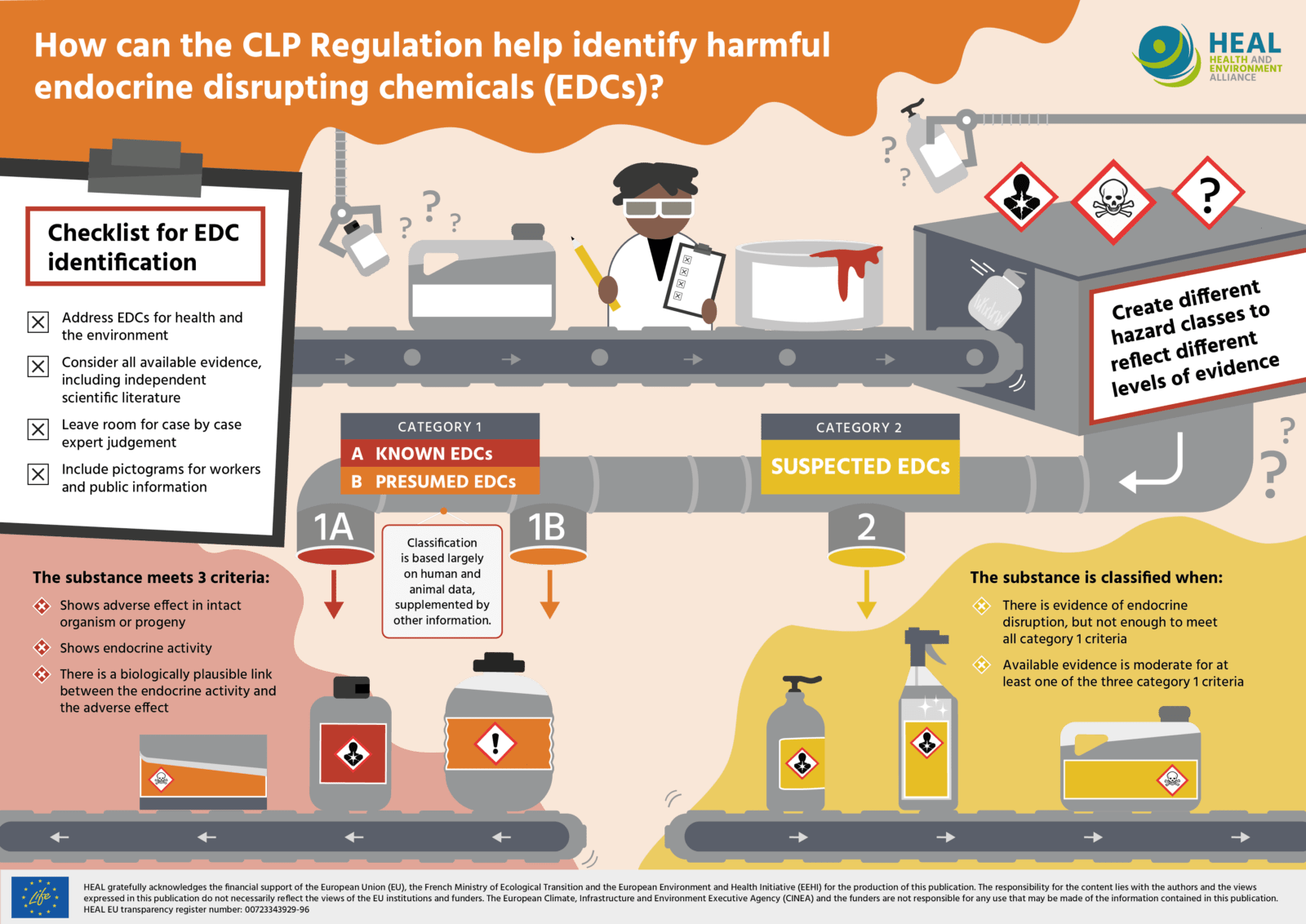
In order to do so, HEAL strongly supports the addition of a hazard class for EDCs in CLP, based on the following principles:
- Address substances with endocrine disrupting properties for both human health as well as the environment.
- Consider and reflect all available evidence, including independent scientific literature.
- Include subcategories in order to reflect the varying levels of scientific evidence in the course of EDC identification. As is the case for carcinogens, mutagens, and reprotoxicants, the categories should allow for identification of ‘known’ endocrine disruptors (category 1 A), ‘presumed’ endocrine disruptors (category 1 B) and ‘suspected’ endocrine disruptors (category 2). The latter category is needed to capture those substances, for which there is some evidence of endocrine disrupting properties, but not sufficiently in order to meet the criteria for category 1.
- Leave room for case-by-case expert judgement, since the identification of endocrine disruptors is a complex process.
- Include pictograms and hazard statements for endocrine disrupting chemicals that are easy to understand for workers and the public. Such pictograms and hazard statements should also reflect the subcategories as described above and the endocrine disruptors’ potential to harm future generations and the environment.
Download our social media assets: